
With the help of Bohr \'s model , calculate the second ionisation energy of helium (energy requi... - YouTube

The first and second ionization potentials of helium atoms are 24.58 eV and 54.4 eV respectively... - YouTube

The ionisation energy of hydrogen atom is 13.6 eV, the ionisation energy of helium atom would be (1988) (a) 13.6 eV (b) 27.2 eV (c) 6.8 eV (d) 54.4 eV

Hydrogen emission spectrum spectroscopy successive ionisation energy patterns related to sub-shells and group of periodic table GCE A Level revision notes
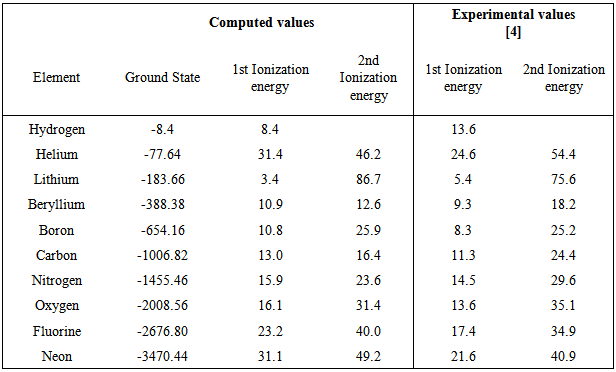
Computation of the First and Second Ionization Energies of the First Ten Elements of the Periodic Table Using a Modified Hartree-Fock Approximation Code

The arrangement of electrons in an atom helps determine the properties and behavior of that atom. - ppt download

Appearance intensity versus ionization energy for nitrogen and helium... | Download Scientific Diagram

If the ionization energy of `He^(+)` is `19.6xx10^(-18) J` per atom then the energy of `Be^(3+)` ion - YouTube










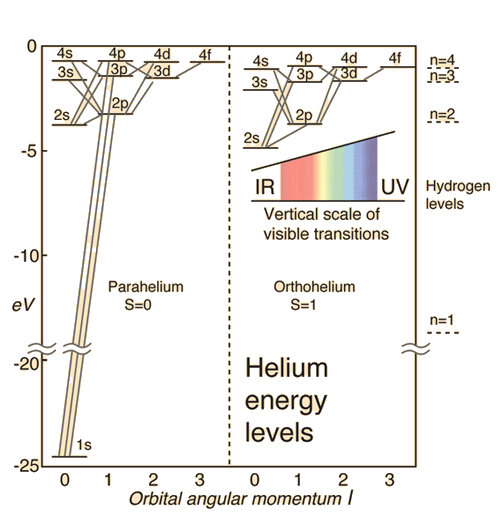

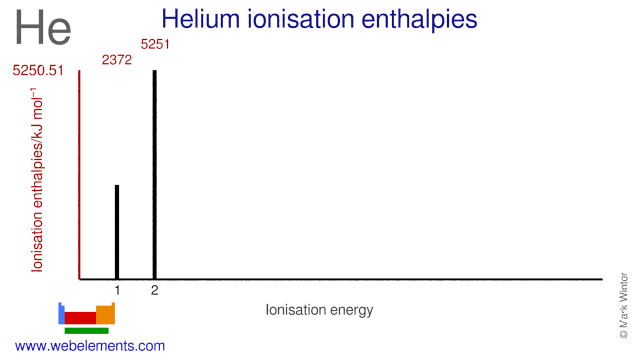
![Ionization energy [eV] of helium atom and molecular ions. | Download Table Ionization energy [eV] of helium atom and molecular ions. | Download Table](https://www.researchgate.net/publication/1790597/figure/tbl2/AS:667036099289106@1536045207928/Ionization-energy-eV-of-helium-atom-and-molecular-ions.png)